Safer CRISPR
Our lab is interested in using the designer DNA binding proteins such as Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) and Zinc Finger proteins to develop novel gene and (epi)-gene editing tools for in vivo therapeutics. We apply design principles of synthetic biology to develop controllable genetic circuits and interface with gene therapies for better controllability and safety upon the clinical translations. In this line, our lab has a 3-tier research program:
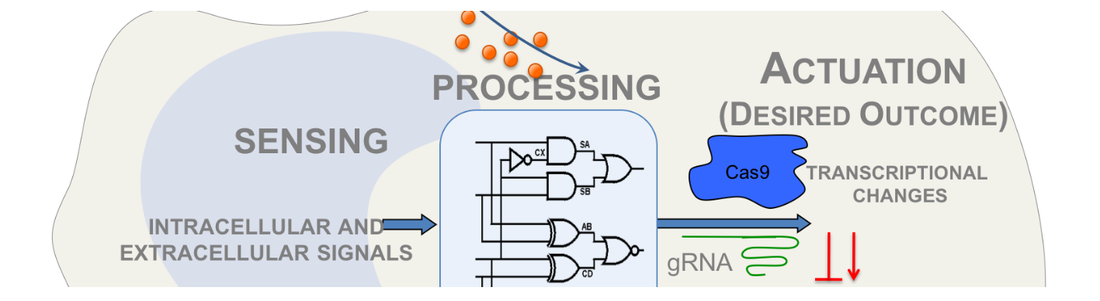
1-Synthetic Biology: We are interested in designing new genetic circuits that confer new behaviors in mammalian cells. Along this notion, we are interested in genetic circuits that can modulate immune response or the genetic circuits that can modulate the function of the gene editors. We apply these tools both in cells and in animal models.
2- In vivo gene and (epi)gene therapies: using different delivery systems such as nanoparticles and Adeno-associated viruses (AAV) we are interested in applying CRISPR-based editors to understand tissue biology and modulate the course of diseases, with specific attention to immune response. We are interested to develop and compare different editors for more efficacious therapies, interested to modulate epigenetic codes to change the course of diseases, and develop strategies to counteract immune reaction against gene editors, viral therapies, infectious viruses or chronic inflammatory conditions.
3- Human microphysiological systems that can report on immune response to and toxicity following the application of gene editors to human tissues are crucial in developing clinical gene editors. We work with Liver on Chip platform, a 3D perfusable system that allows us to develop and maintain human liver tissue over extended period of time. We use patient-specific cells to assemble immune-competent liver tissues in this platform and analyze their response to gene editing events through high throughput assays such as transcriptomics, genomics and Luminex assays. The goal is to develop biomarkers and predict response to gene editing in human tissues.
1-Synthetic Biology: We are interested in designing new genetic circuits that confer new behaviors in mammalian cells. Along this notion, we are interested in genetic circuits that can modulate immune response or the genetic circuits that can modulate the function of the gene editors. We apply these tools both in cells and in animal models.
2- In vivo gene and (epi)gene therapies: using different delivery systems such as nanoparticles and Adeno-associated viruses (AAV) we are interested in applying CRISPR-based editors to understand tissue biology and modulate the course of diseases, with specific attention to immune response. We are interested to develop and compare different editors for more efficacious therapies, interested to modulate epigenetic codes to change the course of diseases, and develop strategies to counteract immune reaction against gene editors, viral therapies, infectious viruses or chronic inflammatory conditions.
3- Human microphysiological systems that can report on immune response to and toxicity following the application of gene editors to human tissues are crucial in developing clinical gene editors. We work with Liver on Chip platform, a 3D perfusable system that allows us to develop and maintain human liver tissue over extended period of time. We use patient-specific cells to assemble immune-competent liver tissues in this platform and analyze their response to gene editing events through high throughput assays such as transcriptomics, genomics and Luminex assays. The goal is to develop biomarkers and predict response to gene editing in human tissues.